The Air Compression Process – What Happens?
The air compression process can be split into six stages:
- compressor OFF just before starting;
- starting the compressor;
- building up pressure in the receiver;
- arriving at the working pressure;
- after some use the temperature of the air in the receiver has risen;
- the pressure in the receiver drops due to high load demand.
Contents
Stage by Stage
During the air compression process this mixture of gases (nitrogen, oxygen, argon, water vapour etc.) undergoes a number of physical changes during the conversion from ambient to pressurized air. The sponge analogy will will help you understand some of these complex changes.
Air Compression Process: Before turning on the compressor
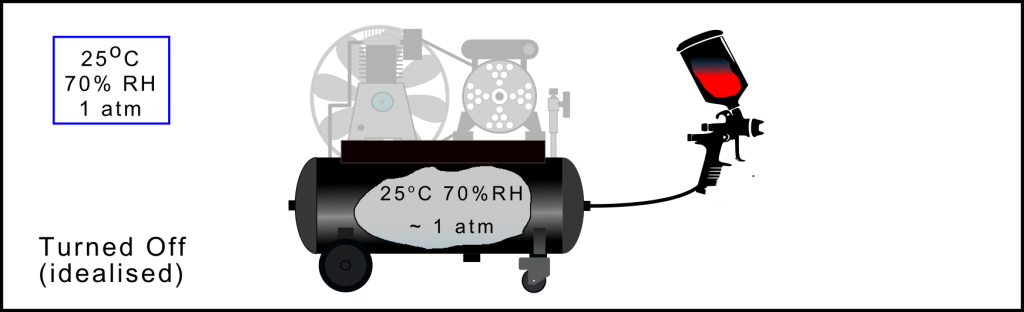
In this example, before starting the compressor, the outside air is at 25oC and has a relative humidity of 70%. The air stored in the receiver is at the same temperature and has the same relative humidity as the ambient air. However, if there were any water in the receiver, pretty much always the case, then the air in this enclosed volume would be saturated (100% RH) and the dew point temperature would be the same as that of the outside air, 25oC.
Air Compression Process: Starting the compressor
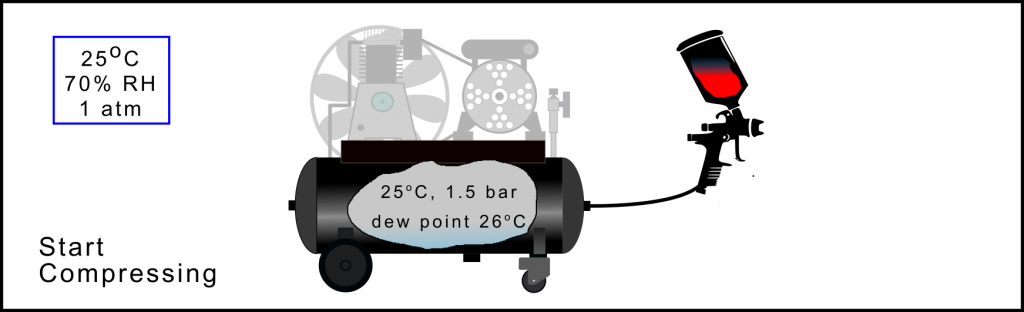
The receiver’s air pressure starts to increase when the compressor is turned on. In this example when the pressure reaches about 1.5 bar(a) or only 0.5 bar above ambient pressure the air inside the receiver has a dew point temperature of ~26oC. In other words the air is saturated. Once the dew point increases above 25oC, the ambient temperature, water vapour in the only slightly compressed air will start to condense within the receiver and form a pool along its base. Obviously the pressure at which the compressed air starts to become saturated depends upon the ambient temperature and the amount of water vapour in the ambient air, its relative humidity. However, it’s very surprising how low this pressure is, see technical section for plots indicating this effect.
Air Compression Process: Increasing the stored pressure
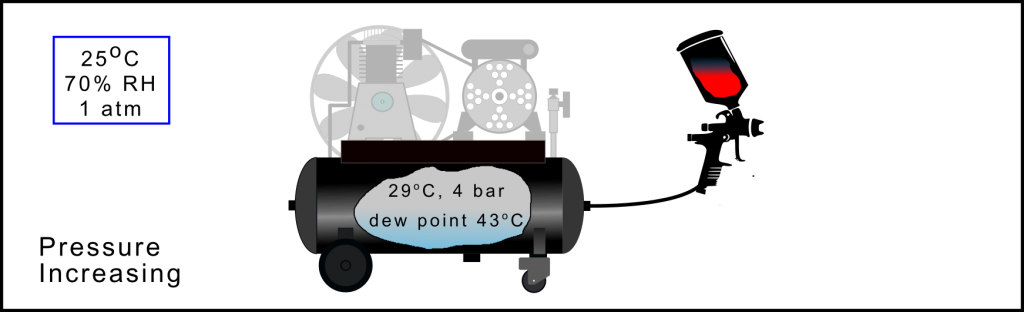
As the pressure of the compressed air further increases (the sponge is squeezed) the dew point temperature rises, an indication that the air is not able to hold as much water vapour. However, as the compressor has now been operational for a short period the temperature of the air in the receiver also starts to rise (increasing the size of the sponge) and counteracting the squeezing effect of pressure. In all operational conditions it is this competing expansion of the sponge, due to increasing temperature, and squeezing of the sponge, due to increasing pressure, that determines the ratio of water in the vapour and liquid phase. N.B. In the above example approximately 45% of the total system water is still in vapour phase within the compressed air.
Air Compression Process: At working pressure
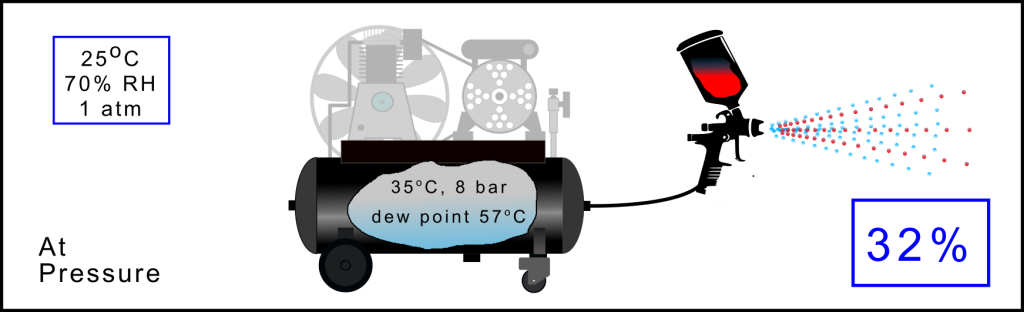
When at working pressure, in this case 8 bar(a), and having a compressed air temperature of 35oC the dew point is 57oC. So for any temperature below this, and the compressed air is only at 35oC, the sponge is too small and water vapour condenses. When used, in this case to spray, about 32% of the total water vapour that was sucked in from the ambient air is still present in the air stream.
Air Compression Process: Temperature increases in the receiver
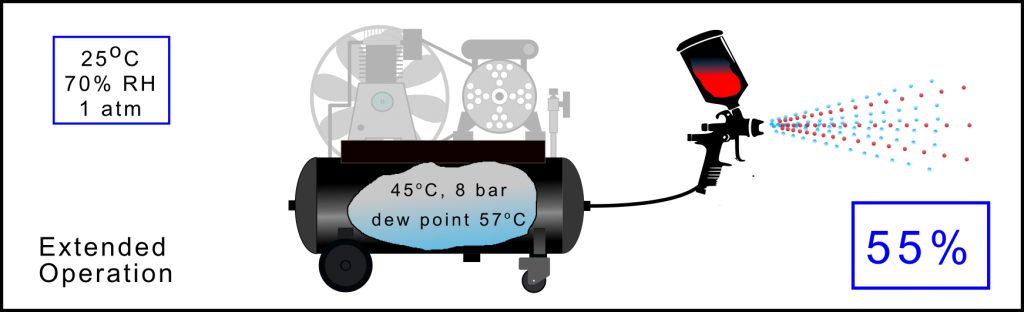
Assuming that the compressor has been extensively used (has a high load factor) then the temperature of the air will slowly rise in the receiver. In this case 10oC when compared with the previous condition. As the temperature has risen the sponge has increased in size and is capable of holding more water vapour. When used under these conditions the amount of water vapour in the compressed air has risen to 55% of the total water drawn in from the ambient air.
As in the previous case, this air is drawn through all parts of the system and will produce water (when the temperature drops) along all the pipework, through the conditioning equipment and at the tool. In addition, cooling of the air as it leaves the tool the so –called Joule-Thomson effect will directly generate water, in this case, into the sprayed finish. Once the compressed air is saturated it will stay saturated throughout the whole air delivery system and continually produce water. This is not good for your tooling and applications.
Air Compression Process: Pressure reduction due to load demand
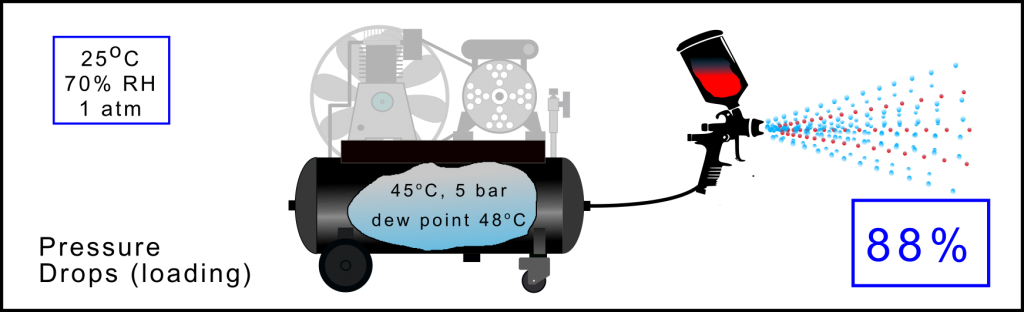
When using small, or under rated, compressors it is unlikely, under sustained load, that the compressor will be able to maintain the required delivery pressure. Say, as in the case above, the receiver pressure starts to drop and reaches 5 bar(a). As the pressure has reduced the sponge has been able to expand and is now capable, under these conditions, of supporting more water vapour. In this case about 88% of the water from the input ambient air is directly pumped through the system and out of the tool. So working for sustained time periods even at low pressures means that significant amount of water is passed through the compressed air system.
Water Generation in the air compression process
In summary:
- Pretty much wherever you are, if you used compressed air over 2 bar you will generate water and water vapour will be present, in varying amounts, in the air that you use.
- Once you have water in your system it is very difficult to remove and will affect the performance of your tools and the quality of the processes that you use.
- Any time you subject your compressor to high load demands that result in a pressure drop at the regulator (this is a very common occurrence with small/medium compressor systems) you are directly pumping additional water and water vapour through your system.
- Even if you trap out liquid water (quite easy to do) your compressed air will still be saturated and the amount of water vapour that comes out at the end will still depend on the temperature of the compressed air and its pressure history.
- To date, there has been no simple, passive means of producing water free compressed air other than resorting to refrigerant and other dryer technologies.
See Also
Dry Tank Technology – Technical Description
Conventional vs DTT Compressor
References
